The latest addition to Shimadzu’s analytical balance line, the AR series features the 4th generation of the patented UniBloc weighing cell, providing fast response times and high stability. The AR balances are fully GLP/GMP compliant and can also support CFR 21 part 11.
AP series
Improved analytical balances with UniBloc load cell

Features
- High weighing speed. Shimadzu analytical balances feature a single-piece UniBloc weighing sensor, which is now even more advanced. The response time for trace measurements (from 1 mg) is reduced to approximately 2 seconds, compared to 10 seconds for the previous model. This significantly improves weighing efficiency.
- A wide range of accessories and options suitable for semi-micro measurements.
- The built-in high-performance STABLO-AP ionizer (optional) eliminates static electricity 1/10 faster than previous models, ensuring reliable measurements with a simpler procedure.
- The AP holder for various containers (test tubes, flasks) is provided as standard.
The combined use of the AP holder and the STABLO-AP ionizer allows you to quickly remove static electricity from the entire test chamber, including the surfaces of glassware, which helps reduce weighing time and increases measurement reliability.
- Regulation for the pharmaceutical industry. A solution to improve operational efficiency and ensure data reliability – the LabSolutions Balance analytical network data system – ensures compliance with various regulations to ensure the integrity of measurement data, including ISO 17025 for testing laboratories, ISO 9001 and ISO 14001 for the manufacturing industry, as well as GLP/GMP and United States Pharmacopeia (USP) for the pharmaceutical industry.
- Excluding manual input, all weighing data can be automatically saved in a secure database without transcription errors.
- Reports suitable for weighing methods such as mass change test, weight loss on drying test and particle size test can be automatically generated after measurement. In addition, customized reports can be generated containing information such as system compliance, content uniformity and elution tests, as well as analysis results obtained by HPLC, etc.
- Complies with the latest recommendations for ensuring data integrity (US FDA 21 CFR Part 11).
- The integrated report generation function combines HPLC analysis results and weighing results from the balance.
- The periodic review support function (APW / APX series only) allows you to check repeatability, angular load error and linearity simply by following the on-screen instructions.
- Buffer solution preparation mode (APW series only). Recipes for 13 commonly used buffer solutions are included as standard and provided as standard. New recipes can be registered independently. If a buffer solution recipe is not registered by default, it can be done again. Instructions are shown on the display. The target weighing value is shown on the display and analog panel for comparison of the target with the current weight. Manual calculation is not required. There is a function to record the output with date, time and operator name.
- Various functions to support the user. The standard configuration includes an RS-232C connector, a USB device, and a USB host (APW models only). You can output data to a PC and a printer simultaneously, or connect a USB flash drive, barcode scanner, or external numeric keypad.
APW models
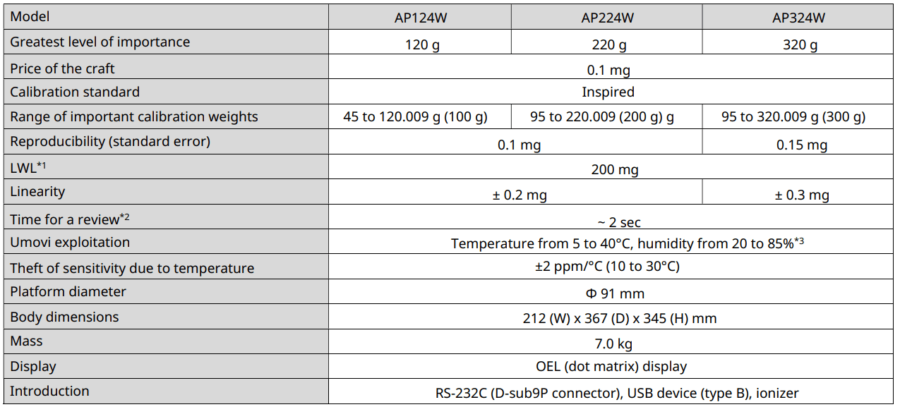
*1 According to USP Capture 41. This is the value calculated by weighing a sample with a mass of 5% of the balance’s LWL.
*2 Typical response time value.
*3 Without condensation.
APX models
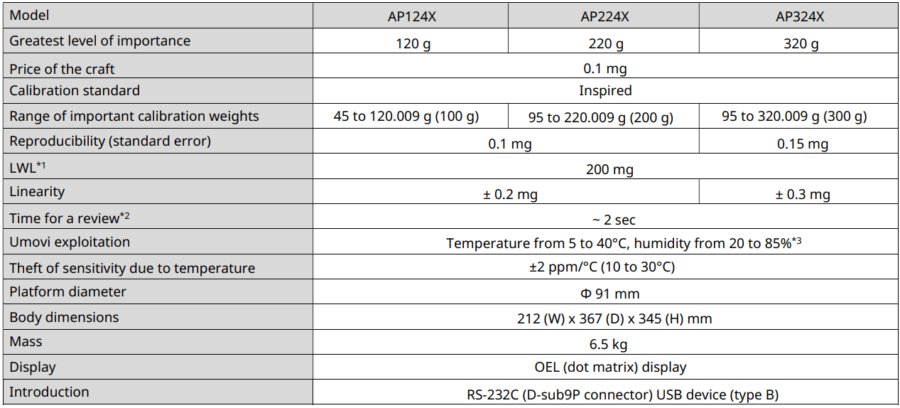
*1 According to USP Capture 41. This is the value calculated by weighing a sample with a mass of 5% of the balance’s LWL.
*2 Typical response time value.
*3 Without condensation.
APY models
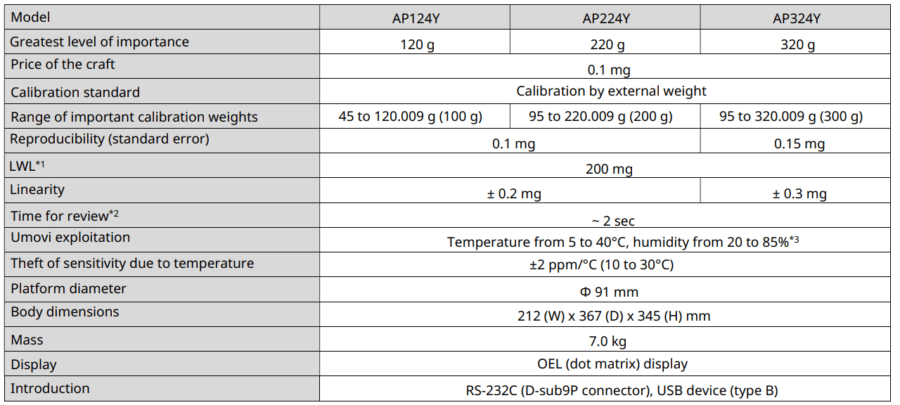
*1 According to USP Capture 41. This is the value calculated by weighing a sample with a mass of 5% of the balance’s LWL.
*2 Typical response time value.
*3 Without condensation.


