Shimadzu’s TOC-1000e is an innovative in-line analyzer for ultrapure water quality control, total organic carbon, specifically designed to address the growing demands applied in water quality control in the pharmaceutical, food, chemical and semiconductor industries.
TOC-1000e

Compactness and efficiency
- The smallest TOC analyzer in its class in the world with a color touch display, which allows you to save workspace.
- The lightweight design (weighing only 2.88 kg) provides high variability in the choice of installation location.
- The compact dimensions (270 x 140 x 180 mm) allow you to install the device in limited space.
Environmental friendliness and innovation
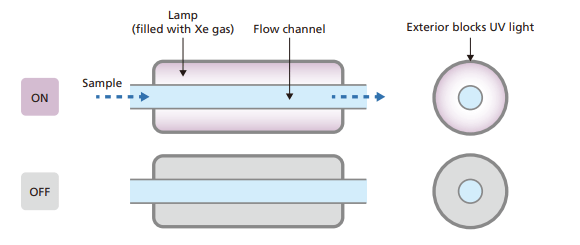
- The world’s first TOC analyzer with a mercury-free excimer lamp, reducing environmental impact.
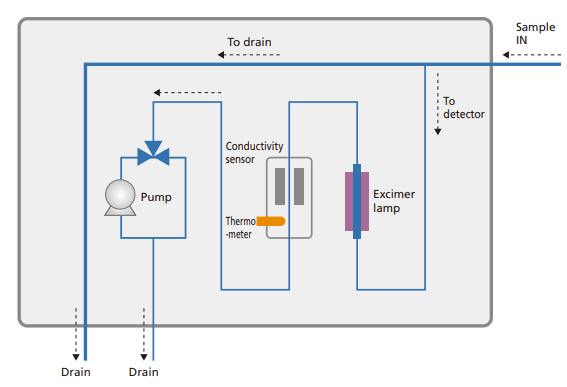
- Shimadzu’s new “Active-Path” technology provides efficient UV oxidation by integrating the sample flow line directly into the lamp.
- The outer material of the lamp blocks UV radiation, preventing ozone formation and increasing safety.
High sensitivity
- The detection limit of 0.1 μg/L enables precise control of ultrapure water.
- The TOC measurement range of 0 to 2000 μg/L meets a variety of analysis needs.
- Compliance with the requirements of the United States and European Pharmacopoeia ensures reliable results for the pharmaceutical industry.
Простота використання та обслуговування
- The intuitive 7-inch color touch display simplifies operation and data reading.
- Annual maintenance without the need for special tools reduces operating costs.
- Convenient calibration and qualification with the optional sampler increase the reliability of the obtained results.
Flexibility in connection and management
- Installation options (desktop, wall or pipe mount) adapt to different laboratory conditions.
- Data export in CSV and PDF formats via USB ensures easy integration with existing data management systems.
- Remote access via web browser allows you to control the instrument from anywhere.
Regulatory compliance
- Security features for FDA 21 CFR Part 11 compliance, including user authentication and event logging.
- Ability to integrate with LabSolutions database for centralized data management from multiple instruments.
- Certified standards available for calibration and system suitability testing.